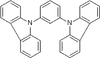1,3-Di-9-carbazolylb
enzene (purified by sublimation) / 1,3-Bis(carbazol-9-y
l)benzene In 1977, Shirakawa and co-workers reported that thin films of the semi-conducting polymer polyacetylene show a dramatic increase in electrical conductivity when doped with controlled amounts of iodine.1) Their reports triggered intensive R&D into the electrical conductivity of plastic materials. Studies of conducting polymers began and, as a result, many ï-conjugated polymers such as polypyrrole2), polythiophene3), polyaniline4) and polyphenylenevinylen
e5) have been developed. Among these polymers, many of them have been utilized practically. One example is an electrolytic condenser using polypyrrole. This condenser has characteristic features such as being compact and lightweight, as well as having high-capacity and high-frequency compliance. These features have achieved a downsizing and weight-saving in electronic devices, and are utilized currently in some mobile phones. Thereby, conducting polymers are widely used for electronic devices vital to our everyday life. These and many other achievements were the reasons Shirakawa received to the Nobel Prize Award in 2000. Moreover, conducting polymers have been applied to solar cell materials. For instance, Kim et al. have reported the synthesis of the co-polymers 3 and 4, using 4,7-dibromo-2,1,3-be
nzothiadiazole (1) and 4,7-dibromo-2,1,3-be
nzoselenadiazole (2) as starting materials, respectively. According to their results, the bulk heterojunction solar cells composed of 3 or 4 with PC71BM give power conversion efficiencies of 1.12%6) and 1.34%7), respectively. Thus, further applications using conducting polymers can be fully expected in many areas.
1,3-Di-9-carbazolylbenzene (purified by sublimation) / 1,3-Bis(carbazol-9-yl)benzene
In 1977, Shirakawa and co-workers reported that thin films of the semi-conducting polymer polyacetylene show a dramatic increase in electrical conductivity when doped with controlled amounts of iodine.1) Their reports triggered intensive R&D into the electrical conductivity of plastic materials. Studies of conducting polymers began and, as a result, many Ï-conjugated polymers such as polypyrrole2), polythiophene3), polyaniline4) and polyphenylenevinylene5) have been developed. Among these polymers, many of them have been utilized practically. One example is an electrolytic condenser using polypyrrole. This condenser has characteristic features such as being compact and lightweight, as well as having high-capacity and high-frequency compliance. These features have achieved a downsizing and weight-saving in electronic devices, and are utilized currently in some mobile phones. Thereby, conducting polymers are widely used for electronic devices vital to our everyday life. These and many other achievements were the reasons Shirakawa received to the Nobel Prize Award in 2000.
Moreover, conducting polymers have been applied to solar cell materials. For instance, Kim et al. have reported the synthesis of the co-polymers 3 and 4, using 4,7-dibromo-2,1,3-benzothiadiazole (1) and 4,7-dibromo-2,1,3-benzoselenadiazole (2) as starting materials, respectively. According to their results, the bulk heterojunction solar cells composed of 3 or 4 with PC71BM give power conversion efficiencies of 1.12%6) and 1.34%7), respectively. Thus, further applications using conducting polymers can be fully expected in many areas.